  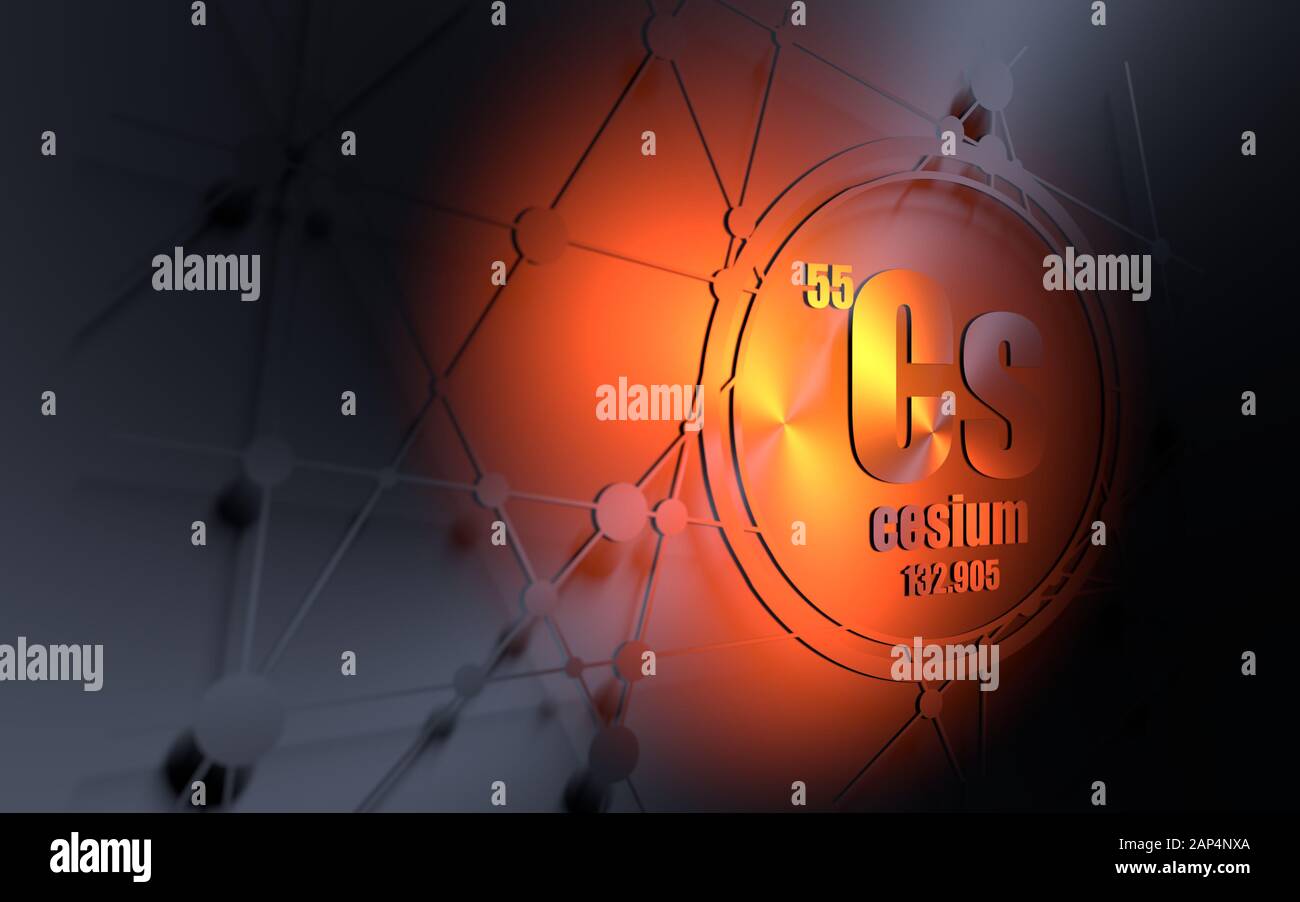
Just the factsĪtomic number (number of protons in the nucleus): 55Ītomic symbol (on the periodic table of elements): CsĪtomic weight (average mass of the atom): 132.90ĭensity: 1.086 ounces per cubic inch (1.879 grams per cubic centimeter) Cesium is a monoisotopic element, and hence the only naturally occurring nuclide 133Cs. Cesium salts color the nonluminous flame of a Bunsen gas burner reddish violet or bluish purple (460 nm). Cesium is the most reactive of the alkali metals with nitrogen, carbon, and hydrogen. Like the other alkali metals it combines vigorously with mercury, forming a stable cesium amalgam. Moreover, cesium hydroxide is the strongest base known. It is readily soluble in liquid and anhydrous ammonia, giving deep blue solutions. Therefore, it must be kept immersed in mineral oil. It ignites spontaneously in oxygen and hence is difficult to handle because it reacts spontaneously in air. It decomposes water vigorously, evolving hydrogen and forming the caustic cesium hydroxide, CsOH, and it even reacts with ice at temperatures above –116☌. 28.4☌), it can be liquid at room temperature. Like the other alkali metals, it has a body-centered cubic crystal lattice structure. It would melt in your hands - if it didn't explode first, as it is highly reactive to moisture.Ĭesium is a soft, malleable, silvery-white element when freshly cut and has the highest density (1873 kg.m–3) of the alkali metals, just slightly greater than that of magnesium. 
Cesium is a rare, silver-white, shiny metal with brilliant blue spectral lines the element's name comes from "caesius," a Latin word meaning "sky blue." It is the softest metal, with a consistency of wax at room temperature. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
